The Alameda County Public Health Department sends out Health Alerts, Advisories, and Updates regarding communicable disease outbreaks, immunization updates, and other public health concerns to Alameda County clinicians and other partners. See recent releases below.
Sign up for

Subscribe today to AC Alert, Alameda County’s 24/7 notification system, to begin receiving emergency alerts.
Health Care Provider AC Alert Registration Form: Health Updates, Advisories, and Alerts, as well as other pertinent information, are sent out to healthcare providers and other relevant contacts in Alameda County through a Public Health-specific subscription in AC Alert. Healthcare providers can register to receive these communications.
Main AC Alert Registration Form: All Alameda County residents are encouraged to register themselves for AC Alert messages from several Alameda County agencies via other subscription options.
All Health Alerts
| ALERT | conveys the highest level of importance; warrants immediate action or attention. |
| ADVISORY | provides important information for a specific incident or situation; may not require immediate action. |
| UPDATE | provides updated information regarding an incident or situation; unlikely to require immediate action. |
July 21, 2025 |
Advisory |
CDPH Mycoplasma pneumoniae Infections Among Children and Adolescents During Fall and Winter Respiratory Illness Season On July 9, 2025, CDPH issued a CAHAN reporting an increasing number of respiratory infections in children due to Mycoplasma pneumoniae (M. pneumoniae) since March 2025.
For more information, please see this PDF from CDPH. |
July 21, 2025 |
Alert |
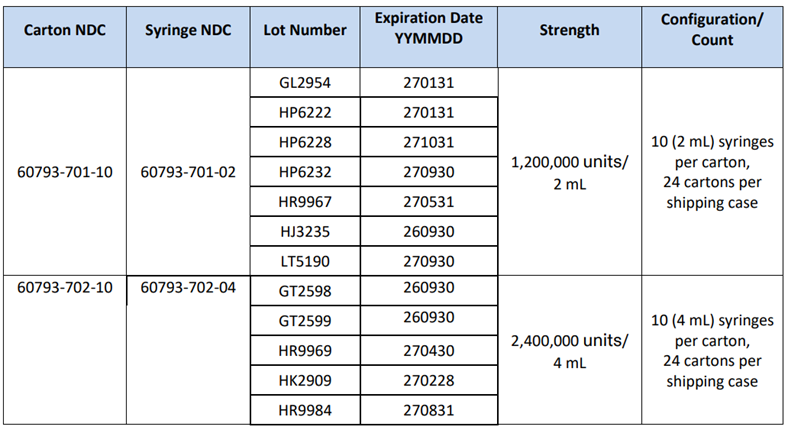
Penicillin G Benzathine (Bicillin® L-A) Voluntary Recall and Shortage July 17, 2025 SITUATION: On July 10, 2025, Pfizer, the manufacturer of penicillin G benzathine (Bicillin® L-A), issued a voluntary recall of affected product lots due to particulates identified in the syringes during visual inspection. This includes both 1.2 million units/2 mL and 2.4 million units/4 mL prefilled syringes distributed from December 11, 2023 – June 24, 2025. Please check your Bicillin stock immediately as affected lots (see below) should be discontinued. You can find more information about credit reimbursement for returned affected lots in Pfizer’s Bicillin® L-A recall notification. In response to this recall and as Pfizer is the only manufacturer of Bicillin® L-A, the FDA anticipates a limited supply of Bicillin® L-A 1.2 and 2.4 million units doses with no estimated recovery date. Bicillin® L-A is the only acceptable treatment for pregnant people infected with or exposed to syphilis and should be prioritized for babies exposed to syphilis in utero. Utilizing Pfizer’s Medical Request Process, providers and pharmacy directors who no longer have wholesale or hospital system access to Bicillin® L-A can submit a medical request form for patients who can only receive Bicillin® L-A due to the risk of congenital syphilis. Doxycycline is an acceptable alternative for non-pregnant adults with primary, secondary, or early and late latent syphilis, and for those who have been exposed to a patient with infectious syphilis. Limited data support ceftriaxone as an effective therapy option for the treatment of primary and secondary syphilis, and neurosyphilis, in non-pregnant adults. Extencilline and Lentocilin were approved by FDA for temporary importation due to prior shortages and remain available. Other intramuscular formulations of penicillin (e.g. Bicillin C-R) are not acceptable alternatives for the treatment of syphilis. ACTIONS REQUESTED OF HEALTHCARE PROVIDERS:
RESOURCES:
|
June 24, 2025 |
Advisory |
CDPH Influenza A Subtype Testing Guidance: Continued Surveillance During the Summer Months On June 11, 2025, CDPH issued a CAHAN urging healthcare providers to maintain awareness of potential H5N1 infection during the summer months. Continued influenza testing and subtyping all flu A positives over the summer is key to maintaining visibility of the flu A subtypes circulating in the community, and for enhanced detection of novel influenza cases. Healthcare providers should:
Clinical and commercial laboratories should: Report any samples that are flu A positive and for which subtyping was attempted but did not identify a seasonal flu A subtype to ACPHD and urgently direct these samples to ACPHL. From June through September 2025, submit flu A positive samples that were not subtyped in clinical or commercial laboratories to ACPHL for subtyping. |
June 09, 2025 |
Advisory |
Ongoing Transmission of Candida auris in Healthcare Facilities in Northern and Central California As an update to the March 2023 Health Advisory describing the emergence of Candida auris (C. auris) in Northern California, the California Department of Public Health (CDPH) and local public health partners are alerting health care providers of an increase in C. auris cases identified across Northern and Central California. In addition, there has been ongoing transmission in acute care hospital and long-term acute care hospital (LTACH) settings in the Bay Area and Central Valley since November 2024, with more spread likely in the region than has been identified to date. Patients and residents who have had prolonged admission in healthcare settings, particularly high-acuity care settings including LTACHs, ventilator-equipped skilled nursing facilities (vSNFs) and high-acuity ACH units (e.g., intensive care units (ICUs)), are at highest risk of C. auris and other multidrug-resistant organism (MDRO) colonization and infection. |
May 20, 2025 |
Advisory |
Transmission of Candida auris at an Alameda County Acute Care Hospital Alameda County Public Health Department (ACPHD) is investigating transmission of Candida auris (C. auris) among patients at Highland Hospital (HH) in Oakland. To date, five cases of C. auris colonization have been identified from multiple adult inpatient units at HH. The HH Emergency Department and Labor & Delivery Unit are not currently affected. Contact tracing and additional point prevalence surveys (PPS) are being conducted. No transmission of C. auris has been documented in any other local facility at this time. ACPHD is notifying local health care facilities to inform actions to prevent further spread (see attached Health Advisory). ACTIONS REQUESTED OF LOCAL HEALTH CARE FACILITIES
ACTIONS REQUESTED OF CLINICAL AND REFERENCE LABORATORIES
|
May 07, 2025 |
Advisory |
Congenital Syphilis Health Advisory This Health Advisory is to update providers on congenital syphilis. Alameda County’s congenital syphilis rate increased 218% between 2019 and 2023. Congenital syphilis is preventable with routine screening and timely treatment of syphilis in pregnant persons. More information on this review can be found here. Congenital Syphilis Health Advisory Actions Requested of Clinicians:
For Pregnant Patients:
For any questions, please contact the ACPHD Sexual Health team Monday through Friday, 8:30 AM – 5:00 PM, at (510) 267-3250 or email us at SexualHealth@acgov.org. |
March 12, 2025 |
Advisory |
Expanding Measles Outbreak in the United States and Guidance for the Upcoming Travel Season The purpose of this advisory is to share the CDC HAN from 3/7/25 about a measles outbreak in Texas and New Mexico, emphasizing the importance of being up-to-date on MMR vaccination. As of March 6, 2025, a total of 222 measles cases had been reported by twelve U.S. jurisdictions this year, including California. Most of the 222 cases are among children who had not received the MMR vaccine. Per Title 17, cases of measles or suspect measles must be immediately reported by telephone, while the patient is still present if possible, to ACPHD at 510-267-3250 (M-F 8:30-5PM). After hours and on weekends, please call the Alameda County Fire Dispatch at 925-422-7595 and ask to speak with the Public Health Duty Officer on-call. Delays in reporting can result in delayed diagnosis and on-going transmission. ACTIONS REQUESTED OF CLINICIANS
For more detailed guidance, please see the full HAN and Clinical Overview of Measles (CDC) |
February 28, 2025 |
Advisory |
Influenza-Associated Encephalopathy and Encephalitis in Children This purpose of this Health Advisory is to inform clinicians about reports of pediatric influenza-associated encephalopathy and encephalitis (IAE), including four deaths nationally from acute necrotizing encephalopathy (ANE), a severe form of IAE. Centers for Disease Control and Prevention (CDC) has requested that local public health be notified about these conditions. ACTIONS REQUESTED OF CLINICIANS
For more information, please review the complete advisory. |
February 06, 2025 |
Advisory |
Ebola Outbreak Caused by Sudan Virus in Uganda The Centers for Disease Control and Prevention (CDC) has issued a Health Alert Network (HAN) Health Advisory about a recently confirmed outbreak of Ebola disease in Uganda caused by the Sudan virus. Currently, no suspected, probable, or confirmed Sudan virus disease (SVD) cases related to this outbreak have been reported in the United States, or outside of Uganda. However, as a precaution and because there are other viral hemorrhagic fever (VHF) outbreaks in East Africa, CDC is sharing best practices for clinical laboratories and healthcare workers. Recommendations for Clinical Laboratory Biosafety can be found in the full advisory: https://www.cdc.gov/han/2025/han00521.html Recommendations for Clinicians (see full Advisory for complete list):
|
December 09, 2024 |
Advisory |
Detection of Highly Pathogenic Avian Influenza (HPAI) A (H5N1) Virus in Alameda County Alameda County Public Health Department (ACPHD) is issuing this Health Advisory to share information and notify clinicians, public health authorities, and the public about influenza A (H5N1), or bird flu. There are ongoing outbreaks of influenza A (H5N1) in dairy cattle and poultry farms in other counties in California. As of December 9, 2024, there are 58 confirmed human cases in the United States, with 32 cases reported in California. The current public health risk for influenza A (H5N1) infection remains low.
|
November 08, 2024 |
Advisory |
Increase in Pertussis Cases More than 60 confirmed or probable cases of Pertussis have been reported in Alameda County during the 2024-2025 school year, along with multiple high school outbreaks. This is an increase compared with recent years. No hospitalizations have been reported. Despite high vaccination rates in our community, any person who has Pertussis-like symptoms should avoid taking care of or spending time with pregnant persons or infants, as Pertussis can cause severe illness or, in rare instances, death. A person infected with Pertussis is considered contagious for 21 days after the onset of cough or until 5 days of antibiotics have been completed. ACTIONS REQUESTED OF CLINICIANS:
|
October 09, 2024 |
Advisory |
First Marburg Virus Disease Outbreak in the Republic of Rwanda The purpose of this advisory is to share a CDC HAN from 10/3/24 about Rwanda’s first confirmed outbreak of Marburg virus disease (MVD). There were 56 cases and 12 deaths reported as of October 8, 2024, including at least 19 cases in healthcare workers. No confirmed cases of MVD related to this outbreak have been reported in the United States or other countries outside of Rwanda to date. Currently, the risk of MVD in the United States is low; however, clinicians should be aware of the potential for imported cases. Please review the HAN for a full list of recommendations for clinicians, including infection prevention considerations. Contact ACPHD immediately (510-267-3250 M-F 8:30-5PM or 925-422-7595 after hours and on weekends) about any patient who has been to an area with an active MVD outbreak in the past 21 days, AND who has compatible symptoms (e.g., fever, headache, muscle and joint pain, fatigue, loss of appetite, gastrointestinal symptoms, or unexplained bleeding), AND has reported epidemiologically compatible risk factors within the 21 days before symptom onset including direct contact with a symptomatic person with suspected or confirmed MVD, or with any objects contaminated by their body fluids. Recommendations for Infection Prevention and Control Measures in Hospitals
|